Our research interests lie in the field of environmental physical chemistry with emphasis on a detailed molecular-level understanding of complex natural processes and solvation chemistry. To unravel the mechanisms of chemical reactions occurring in a solvent, we employ clusters, free subnanometer-sized particles with well-defined compositions.
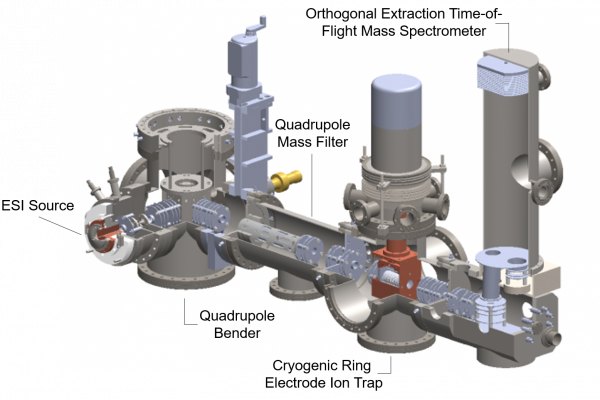
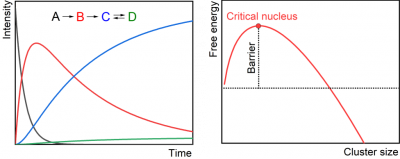
We are developing a powerful ion beam instrument – in-house built tandem mass spectrometer – to investigate particle nucleation and reactivity. The primary goal is to understand the effects of particle size and hydration on new particle formation, identifying critical sizes and stabilization mechanisms. The subnanometer sized particles are generated in a nanoelectrospray source, mass selected in a quadrupole mass filter, and exposed to nucleating precursors under multi-collision conditions in a cryogenic ring electrode ion trap. The reaction time is systematically varied and products of consecutive reactions are analyzed in an orthogonal time-of-flight spectrometer with a reflectron. Our ambition is to provide size and composition selective kinetic data of the nucleation, which also requires the development of dedicated instrumentation.